Key Points
- Injectable semaglutide is available as Ozempic (for type 2 diabetes) and Wegovy (for obesity and cardiovascular prevention), the same active ingredient at different doses for different indications.
- The SELECT trial demonstrated a 20% reduction in major cardiovascular events with Wegovy in overweight or obese patients with established heart disease, even without diabetes. This was a landmark finding that changed how we think about obesity medicines.
- Beyond cardiovascular prevention, injectable semaglutide has demonstrated meaningful benefits in heart failure with preserved ejection fraction obstructive sleep apnoea and chronic kidney disease expanding its clinical relevance significantly.
- These are medicines for select patients with specific indications not general weight loss medicines for the wider population. The decision to prescribe always involves careful clinical assessment.
- Cost and access are real barriers Wegovy in particular is not subsidised in many health systems for the weight/cardiovascular indication, and the annual cost without subsidy is substantial.
- Side effects, particularly gastrointestinal, are common early in treatment but generally manageable with slow dose escalation. These medicines require long-term commitment, the benefits are largely lost if treatment is stopped.
The past three years have seen injectable semaglutide transition from a well-regarded diabetes medicine to one of the most clinically significant developments in cardiovascular prevention in recent memory. The evidence has accumulated rapidly, from heart attack prevention to heart failure, sleep apnoea to kidney disease, and with it, a great deal of public attention, genuine excitement, and some important questions about who these medicines are really for.
This article covers the full injectable semaglutide story, Ozempic and Wegovy, the major clinical trials, the genuine benefits, the honest limitations, and the access conversation that cannot be separated from the clinical one.
For patients specifically interested in the tablet form, Rybelsus, see our dedicated article: Oral Semaglutide: What You Should Know About the Pill Form of Ozempic.
Ozempic and Wegovy, The Same Medicine, Different Doses, Different Roles
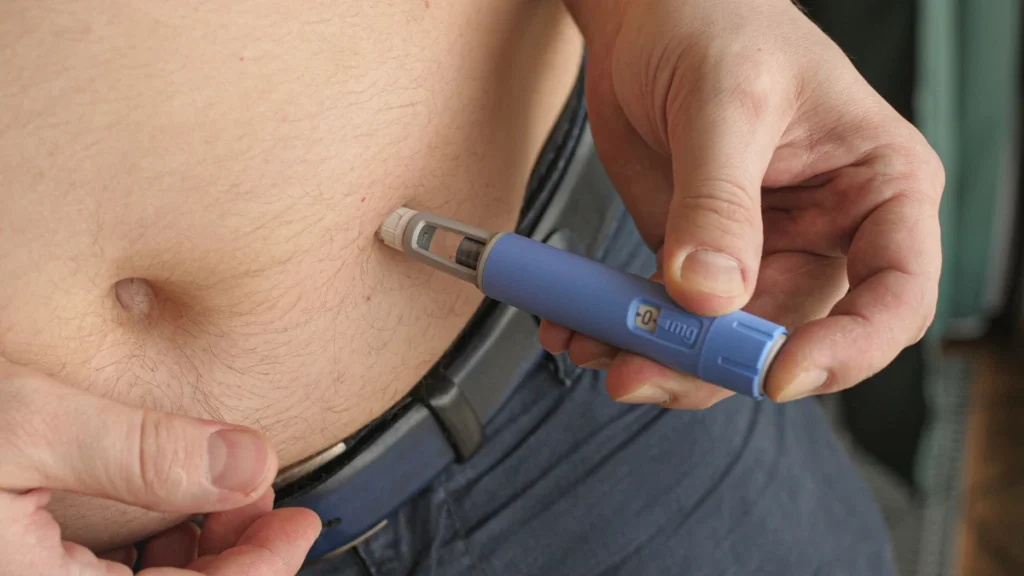
Both Ozempic and Wegovy contain semaglutide, a GLP-1 receptor agonist that mimics a natural gut hormone released after eating. By activating GLP-1 receptors in the pancreas and brain, semaglutide stimulates insulin release in response to glucose, suppresses the appetite-raising hormone glucagon, slows gastric emptying, and reduces appetite through direct effects on the brain’s hunger centres.
| Ozempic | Wegovy | |
|---|---|---|
| Doses | 0.25mg, 0.5mg, 1mg, 2mg weekly | Up to 2.4mg weekly, higher than Ozempic |
| Primary indication | Type 2 diabetes | Obesity / weight management; cardiovascular risk reduction |
| Cardiovascular approval | Yes, in type 2 diabetes with established CVD (SUSTAIN-6) | Yes, in obesity with established CVD without requiring diabetes (SELECT) |
| Average weight loss | Approximately 5–7kg at therapeutic doses | Approximately 15% of body weight at 2.4mg |
| Administration | Weekly subcutaneous injection, abdomen, thigh, or upper arm | Weekly subcutaneous injection, same technique |
The distinction matters clinically. Ozempic at lower doses is primarily a diabetes medicine that also protects the heart. Wegovy at the higher 2.4mg dose is primarily a weight management medicine that also protects the heart, and crucially, its cardiovascular benefit has been demonstrated independently of diabetes, in patients who are simply overweight or obese with existing cardiovascular disease.
The Major Trials, What the Evidence Actually Shows
SELECT, The Landmark Cardiovascular Trial
The SELECT trial is the most important study in this space and the one that genuinely changed the conversation. It enrolled 17,604 patients aged 45 or older across 41 countries, all overweight or obese (BMI 27 or above), all with established cardiovascular disease, and critically, none with type 2 diabetes. Half received Wegovy 2.4mg weekly; half received placebo. Both groups continued their standard cardiovascular medicines throughout.
What made SELECT remarkable was not just the magnitude of benefit but what it implied: the cardiovascular protection was not simply a consequence of better glucose control, because these patients did not have diabetes. The benefit likely comes through multiple pathways, weight loss itself, reduced inflammation, lower blood pressure, improved lipid profile, and potentially direct effects of GLP-1 receptor activation on the heart and vessels.
This was the evidence that led the FDA to approve Wegovy specifically for cardiovascular risk reduction in March 2024, a genuinely new indication for a weight management medicine.
SUSTAIN-6, Cardiovascular Protection in Type 2 Diabetes
Earlier and equally important, the SUSTAIN-6 trial established cardiovascular benefit for injectable semaglutide in patients with type 2 diabetes at high cardiovascular risk, demonstrating a significant reduction in major cardiovascular events compared to placebo on top of standard care. Combined with the SOUL trial data for oral semaglutide, the cardiovascular protection of the GLP-1 class in diabetes is now very well established.
Beyond the Heart, Expanding Evidence
Heart Failure With Preserved Ejection Fraction (HFpEF)
One of the most clinically exciting areas of emerging evidence is in heart failure with preserved ejection fraction a form of heart failure where the heart squeezes normally but does not relax properly, strongly associated with obesity. This condition affects millions of patients globally and has historically been very difficult to treat, few medicines have shown meaningful benefit.
The STEP-HFpEF trial compared semaglutide against placebo in obese patients with this condition. Results showed significant improvements in symptoms, exercise capacity, quality of life, and body weight. For a patient population with very limited therapeutic options, this is clinically meaningful. For more on heart failure treatment, see our article on Entresto and heart failure, another important medicine in this space.
Obstructive Sleep Apnoea
The SURMOUNT-OSA trial demonstrated that semaglutide significantly reduced the severity of obstructive sleep apnoea in obese patients, reducing breathing interruptions per hour by approximately 50% in some groups. Since OSA is independently associated with cardiovascular risk, hypertension, and poor metabolic health, and since obesity is its primary driver, this finding adds a meaningful dimension to semaglutide’s potential benefit in complex patients.
Chronic Kidney Disease
The FLOW trial showed that semaglutide meaningfully reduced the risk of serious kidney disease progression in patients with type 2 diabetes and chronic kidney disease. Given the close relationship between kidney health and cardiovascular risk, this is another important dimension, particularly relevant given that SGLT2 inhibitors also provide significant kidney protection and are frequently used alongside GLP-1 agonists in complex patients. See our article on SGLT2 inhibitors for more on this complementary class.
Who Is This Medicine Really For?
This question matters enormously, because the public conversation around these medicines has significantly outpaced the clinical evidence for many of the people asking about them.
Patients With the Strongest Evidence for Benefit
- Type 2 diabetes with established cardiovascular disease or high cardiovascular risk the evidence from SUSTAIN-6 and SOUL is compelling and guideline-supported. Ozempic is typically the relevant product here.
- Obesity or overweight with established cardiovascular disease the SELECT trial directly supports Wegovy in this group, regardless of whether diabetes is present.
- Obesity with heart failure with preserved ejection fraction emerging but meaningful evidence from STEP-HFpEF, particularly where few other options are effective.
- Obese patients with significant obstructive sleep apnoea SURMOUNT-OSA data supports benefit in this group.
- Type 2 diabetes with chronic kidney disease FLOW trial data supports use alongside or instead of other kidney-protective medicines.
Who These Medicines Are Generally Not For
- People without diabetes, obesity, or established cardiovascular disease seeking general weight loss the risk-benefit calculation is very different in lower-risk individuals. These are powerful medicines with real side effects and significant costs, not lifestyle aids.
- Type 1 diabetes not indicated.
- Personal or family history of medullary thyroid carcinoma or MEN2 absolute contraindication.
- History of pancreatitis requires careful specialist assessment before use.
- Pregnancy must be stopped before conception. Women of childbearing age need effective contraception during treatment and should discuss cessation planning with their doctor.
- Patients unable to sustain the cost long-term the benefits largely reverse when treatment is stopped. Starting a medicine that cannot be maintained financially is not a sound clinical decision.
Side Effects, What to Expect
The side effect profile is real and should be discussed honestly before starting. Gastrointestinal symptoms are the most common reason patients reduce doses or stop treatment in clinical trials.
Common Side Effects
- Nausea the most common side effect, particularly in the first weeks and after each dose increase. Almost universal to some degree but ranges from mild to significant. Eating smaller, lower-fat meals, avoiding eating too quickly, and not lying down immediately after eating all help. Slow dose escalation, starting at 0.25mg and increasing gradually over months, is essential and substantially reduces this.
- Vomiting and diarrhoea less common than nausea but can occur, particularly in early weeks. Stay well hydrated.
- Constipation can alternate with looser stools. Adequate hydration and fibre intake help.
- Reduced appetite a therapeutic effect, but patients must maintain adequate nutrition, particularly protein and micronutrients, even with reduced appetite.
- Muscle loss significant weight loss of any cause can reduce lean muscle mass alongside fat. Maintaining protein intake and resistance exercise during treatment is important and an active area of research.
- Gallstones rapid weight loss increases gallstone risk. Report any upper right abdominal pain to your doctor.
- Injection site reactions generally mild, rotating injection sites helps.
- Fatigue particularly in early weeks, usually settles.
The Cost and Access Conversation
This cannot be separated from the clinical discussion, and it is a conversation that deserves honesty rather than being glossed over.
Wegovy is expensive. Without subsidy, annual costs run into thousands of dollars in most markets, a figure that excludes many patients who might clinically benefit. The subsidy situation varies considerably by country and indication.
In Australia, Ozempic is PBS-subsidised for type 2 diabetes in patients meeting specific criteria. Wegovy for the obesity and cardiovascular indication is not currently PBS-listed meaning patients accessing it for weight management or cardiovascular prevention without diabetes face significant out-of-pocket cost. This creates a frustrating disparity: a patient with diabetes and obesity may access semaglutide affordably, while an equally eligible patient with obesity and heart disease but no diabetes cannot.
Practical Points on Access and Cost
- Check your eligibility for subsidy with your doctor eligibility criteria change as evidence evolves and guidelines update. What is not subsidised today may be in twelve months.
- Supply has been a real issue global demand has at times outpaced manufacturing capacity, affecting patients with diabetes who rely on Ozempic. This is improving but worth discussing with your doctor and pharmacist.
- Generic semaglutide is not yet available in most markets, patent protection runs for several more years. Compounded versions have circulated during shortage periods but carry quality and safety concerns.
- Long-term commitment is required the cardiovascular and metabolic benefits largely reverse when treatment is stopped. If cost means the medicine cannot be sustained long-term, this is a genuine clinical consideration that should be part of the prescribing conversation.
- Advocate for yourself if you believe you have a strong clinical case for subsidised access, discuss it with your specialist. Guidelines are evolving rapidly and your doctor may be able to access government programs or trials.
Injectable Semaglutide as Part of a Broader Plan
These medicines work best as part of a comprehensive approach, not as standalone solutions. For patients with diabetes and cardiovascular disease, injectable semaglutide sits alongside statins, ACE inhibitors or ARBs, antiplatelets, and increasingly SGLT2 inhibitors, each addressing different aspects of cardiovascular risk simultaneously.
Lifestyle measures remain the foundation, diet, physical activity, not smoking, managing blood pressure and cholesterol. Semaglutide can be a powerful addition to that framework for the right patient, but it is an addition, not a replacement. The patients who do best on these medicines are typically those who also embrace lifestyle changes alongside treatment, using the reduced appetite and improved metabolic state the medicine provides as an opportunity to build healthier habits that can persist.
Conclusion
Injectable semaglutide represents one of the most significant advances in cardiovascular prevention in the past decade. The SELECT trial alone, demonstrating a 20% reduction in major cardiovascular events in obese patients without diabetes, would be remarkable by any standard. Combined with the evidence in heart failure, sleep apnoea, and kidney disease, the clinical landscape for this class of medicines is expanding rapidly.
But the public enthusiasm for these medicines has run significantly ahead of their appropriate clinical use, and it is important to be clear: they are not weight loss medicines for the general population, and they are not without real side effects, real costs, and real limitations. For the right patient, with the right clinical indication, the right support, and ideally the right access to subsidised treatment, they can make a meaningful difference to both quality of life and long-term cardiovascular outcomes.
If you think you might be a candidate for injectable semaglutide, the conversation starts with your GP or cardiologist, who can assess whether your specific clinical situation aligns with the evidence, what formulation and dose would be appropriate, and what your realistic access to subsidised treatment looks like.