Key Points
- Cardiac amyloidosis occurs when misfolded proteins accumulate in the heart wall, causing it to stiffen and struggle to pump blood effectively.
- It is more common than previously thought, and for years was significantly underdiagnosed because its symptoms mimic other forms of heart disease.
- Two main types affect the heart: AL amyloidosis (caused by abnormal antibody proteins) and ATTR amyloidosis (caused by a liver transport protein).
- Effective treatments now exist for both types. Early diagnosis is the key, the sooner treatment begins, the better the outcome.
Until recently, a diagnosis of cardiac amyloidosis was often delivered with little hope attached to it. The condition was real, the mechanisms were understood, but the treatments were limited, and clinicians in many parts of the world viewed it as a diagnosis of academic interest rather than clinical urgency.
That has changed dramatically. The approval of effective targeted therapies, including Tafamidis for ATTR amyloidosis and Daratumumab-based regimens for AL amyloidosis, has transformed cardiac amyloidosis from a condition managed palliatively into one that can be meaningfully treated. Survival has improved. Quality of life has improved. And with that shift has come a new imperative: find it earlier.
This article explains what cardiac amyloidosis is, how it presents, how it is diagnosed, and what treatment now looks like in Australia.
Amyloidosis, A Disease of Protein Misfolding
To understand amyloidosis, it helps to start with how proteins normally work. Your body continuously produces proteins, precisely folded molecules that carry out essential functions throughout every organ and tissue. Once they have done their job, they are broken down and recycled in an orderly process.
Amyloidosis describes what happens when this system breaks down. An abnormal protein misfolds, taking on the wrong shape, and instead of being cleared normally, it aggregates into resistant strands called fibrils. These fibrils are extraordinarily difficult for the body to break down. They accumulate progressively in organs, and as they do, they disrupt normal tissue architecture and function.
Which organ is most affected depends on which protein is misfiring, and that determines the type of amyloidosis, the symptoms, and the treatment approach.
The Two Main Types That Affect the Heart
There are many forms of amyloidosis, but two are responsible for the overwhelming majority of cardiac cases:
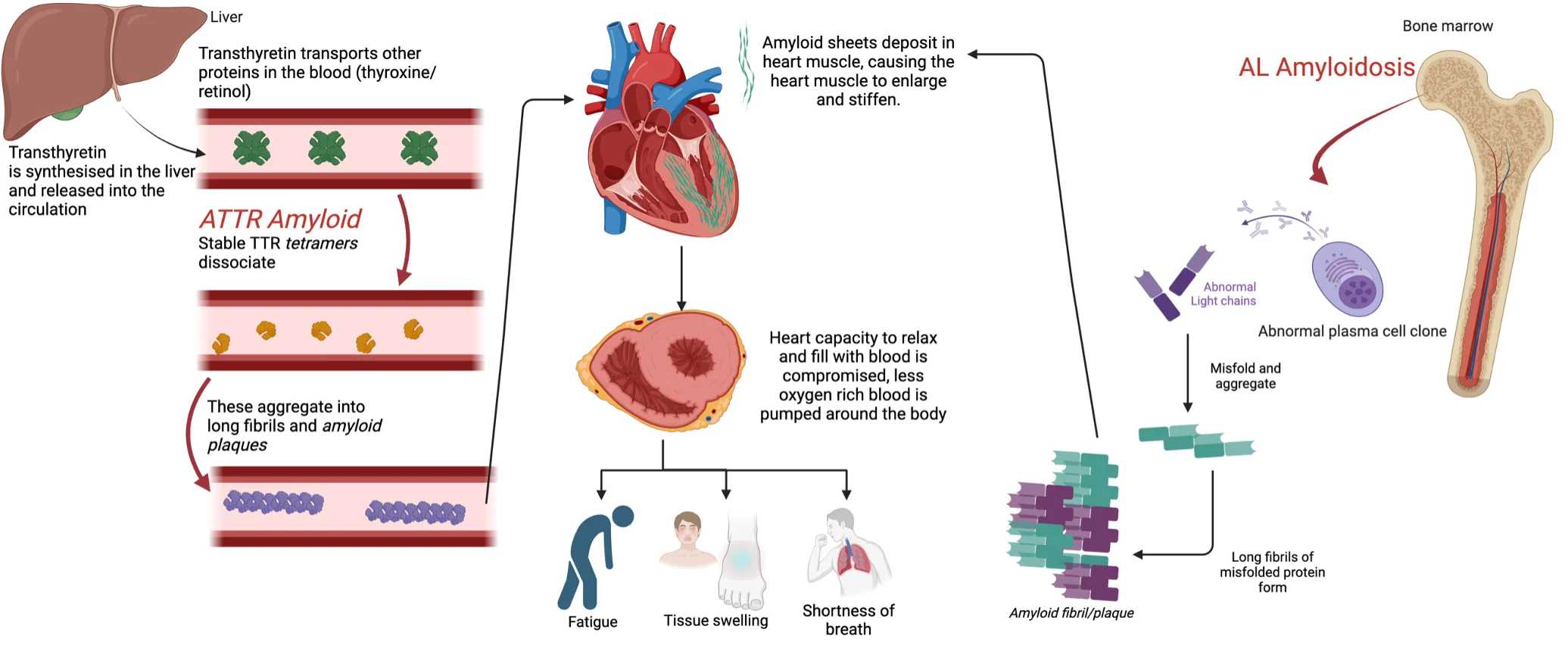
AL amyloidosis (Light-chain amyloidosis) is caused by abnormal plasma cells in the bone marrow producing misfolded light-chain proteins, a component of antibodies. These proteins deposit throughout the body, with the heart and kidneys being the most commonly and severely affected organs. AL amyloidosis is a haematological condition at its root, requiring close collaboration between cardiologists and haematologists.
ATTR amyloidosis (Transthyretin amyloidosis) is caused by misfolding of transthyretin, a transport protein produced by the liver that normally carries thyroid hormone and vitamin A through the bloodstream. ATTR amyloidosis comes in two forms: a hereditary variant caused by a gene mutation, and a wild-type (non-hereditary) variant that occurs as a consequence of ageing. Wild-type ATTR amyloidosis is increasingly recognised as a significant and underdiagnosed cause of heart failure in older adults, particularly men over 65.
new AL amyloidosis cases diagnosed annually in Australia
of heart failure with preserved ejection fraction patients have ATTR amyloidosis
types now have approved targeted therapies available in Australia
How Amyloid Affects the Heart
When amyloid fibrils deposit in cardiac tissue, they infiltrate the walls of the heart, primarily the left ventricle, causing them to thicken and lose their normal elasticity. The result is a heart that cannot relax properly between beats.
This is a specific form of heart failure known as heart failure with preserved ejection fraction (HFpEF) sometimes called “diastolic heart failure.” The heart pumps with normal or near-normal force, but it struggles to fill adequately because the stiffened walls resist expansion. Less blood in means less blood out, and the body’s tissues and organs begin to receive inadequate oxygen delivery.
Cardiac amyloidosis is one of the most important, and historically most overlooked, causes of a stiff heart. Recognising it earlier is now one of the most important things we can do for patients with unexplained heart failure.
Recognising the Symptoms
The symptoms of cardiac amyloidosis develop gradually and are often non-specific, meaning they can easily be attributed to other, more common conditions. This is the primary reason diagnosis is so frequently delayed.
Symptoms that should raise the possibility of amyloidosis include:
- Breathlessness particularly on exertion, but progressing to breathlessness at rest in more advanced disease
- Severe fatigue disproportionate to activity level
- Ankle and leg swelling caused by fluid accumulation as the heart struggles to maintain adequate circulation
- Fainting or near-fainting particularly when standing, due to difficulties maintaining blood pressure
- Numbness, tingling or burning in the hands or feet indicating nerve involvement, which is common particularly in ATTR amyloidosis
- Frothy urine suggesting significant protein loss through the kidneys, more characteristic of AL amyloidosis
It is also worth knowing that amyloidosis is sometimes picked up incidentally, on a routine echocardiogram ordered for another reason, or when a cardiologist notices characteristic features on an ECG or imaging study. Regular check-ups matter.
When Should Amyloidosis Be Considered?
Clinicians are increasingly trained to consider amyloidosis in specific clinical contexts. The four most common scenarios that should prompt investigation are:
- A thickened or stiff heart on echocardiogram with unexplained heart failure, particularly when the walls appear unusually bright or “sparkling”
- Significant protein loss in the urine (nephrotic syndrome) without another clear cause
- Numbness, tingling, or pain in the hands or feet (peripheral neuropathy) alongside heart symptoms
- Difficulty maintaining blood pressure when moving from lying to standing (orthostatic hypotension), causing dizziness or falls
Carpal tunnel syndrome, particularly bilateral carpal tunnel requiring surgery, is also increasingly recognised as an early manifestation of ATTR amyloidosis that can precede cardiac symptoms by years.
How Is Cardiac Amyloidosis Diagnosed?
Diagnosis involves a combination of imaging, blood and urine tests, and in some cases tissue sampling. The approach differs somewhat depending on which type of amyloidosis is suspected.
Echocardiogram cardiac ultrasound remains the cornerstone of initial assessment. In amyloidosis, it typically shows thickened heart walls, impaired relaxation, and characteristic patterns of abnormal muscle appearance. It guides suspicion but cannot confirm the diagnosis alone.
Technetium pyrophosphate (PYP) bone scan a nuclear medicine scan that has transformed the diagnosis of ATTR amyloidosis. Transthyretin amyloid fibrils avidly take up the tracer used in this bone scan, producing a strongly positive result that is highly specific for ATTR cardiac amyloidosis. In the right clinical context, a positive PYP scan can diagnose ATTR amyloidosis without the need for a biopsy.
Blood and urine testing serum free light chains and urine protein electrophoresis are used to identify the abnormal proteins characteristic of AL amyloidosis. These must be checked before a PYP scan is interpreted, as AL amyloidosis can occasionally produce a false positive result.
Bone marrow biopsy required for the diagnosis of AL amyloidosis to identify the abnormal plasma cell clone responsible for producing the misfolded light chains.
ECG and cardiac MRI both provide additional diagnostic information. The ECG in amyloidosis often shows low voltage despite thick walls, a paradoxical finding that is a useful diagnostic clue. Cardiac MRI with gadolinium contrast shows characteristic patterns of late gadolinium enhancement that support the diagnosis.
Treatment, A Rapidly Evolving Landscape
The primary goal of treatment in amyloidosis is to stop further amyloid deposition, giving the body’s natural clearance mechanisms a chance to reduce existing deposits, and preventing further organ damage. Treatment is type-specific and requires close coordination between haematology and cardiology.
Tafamidis, for ATTR Cardiac Amyloidosis
Tafamidis is a landmark therapy for ATTR amyloidosis affecting the heart. It works by stabilising the transthyretin protein in its normal configuration, preventing it from misfolding and aggregating into amyloid fibrils. The pivotal ATTR-ACT trial demonstrated that Tafamidis significantly reduced cardiovascular mortality and hospitalisations compared to placebo in patients with ATTR cardiac amyloidosis. It is now approved in Australia for this indication and represents a genuine advance in the management of a condition that previously had no disease-modifying treatment.
Patisiran, for Hereditary ATTR Amyloidosis
For patients with the hereditary form of ATTR amyloidosis, caused by a genetic mutation in the transthyretin gene, Patisiran offers a different mechanism of action. It is an RNA interference (RNAi) therapy that targets the liver cells responsible for producing transthyretin, dramatically reducing the amount of the protein available to misfold. By lowering the substrate, it reduces amyloid formation throughout the body.
Daratumumab, for AL Amyloidosis
AL amyloidosis requires a fundamentally different treatment approach because the problem originates in the bone marrow. Daratumumab, an antibody that targets and depletes abnormal plasma cells, used in combination with chemotherapy (typically bortezomib, cyclophosphamide and dexamethasone) has become the standard of care for newly diagnosed AL amyloidosis. By eliminating the plasma cells producing the misfolded light chains, this regimen can achieve deep haematological responses and, in many patients, allow existing amyloid deposits to be partially cleared by the body over time.
Ongoing Research and Clinical Trials
The field of amyloidosis research is advancing rapidly. Gene silencing therapies, novel small-molecule stabilisers, and agents designed to actively dissolve existing amyloid deposits are all in various stages of clinical investigation. For patients who do not achieve adequate responses to standard treatments, clinical trials represent an important pathway to accessing emerging therapies. The Australian Amyloidosis Network (AAN) maintains a comprehensive list of active clinical trials and the centres conducting them.
Why Early Diagnosis Matters
The treatments available today work best when started early, before the heart has sustained significant irreversible damage. Delay is not a neutral choice. In symptomatic severe amyloidosis, the prognosis without treatment deteriorates rapidly.
Awareness and self-advocacy are critical. The symptoms of cardiac amyloidosis develop gradually and overlap with more common conditions, making it easy for both patients and clinicians to attribute them to ageing, deconditioning, or other cardiac diagnoses. If you or a family member has unexplained heart failure, particularly with any of the features described above, it is entirely reasonable to ask whether amyloidosis has been considered and whether appropriate investigations have been performed.
The Australian Amyloidosis Network is an excellent resource for patients and families, providing information on symptoms, available treatments, specialist centres, and clinical trials across Australia.
Conclusion
Cardiac amyloidosis has been transformed, from a diagnosis that offered little beyond symptom management to a treatable condition with meaningful therapies and improving outcomes. The challenge that remains is finding it earlier. For patients with unexplained heart failure, breathlessness, or the constellation of features described in this article, pursuing the diagnosis is not just reasonable, it is increasingly urgent. The treatments are there. The diagnosis needs to be made in time to use them.
References
- AbouEzzeddine OF, Davies DR, Scott CG, et al. Prevalence of Transthyretin Amyloid Cardiomyopathy in Heart Failure With Preserved Ejection Fraction. JAMA Cardiology. 2021;6(11):1267–74.
- Wisniowski B, McLeod DSA, Adams R, et al. The epidemiology of amyloidosis in Queensland, Australia. Br J Haematol. 2019;186(6):829–36.