- A myocardial bridge, often just called a muscle bridge, is a common and usually harmless variant present from birth, where a short stretch of a coronary artery dips down and runs through the heart muscle instead of sitting on the surface. Most people who have one will never know and never need treatment.
- In a minority of people, a bridge can cause genuine symptoms, most often chest pain or tightness brought on by exertion or stress. These patients deserve to be taken seriously rather than reassured by default.
- A standard angiogram often underestimates a bridge. Confirming whether a bridge is actually causing symptoms usually needs more detailed assessment, including intravascular ultrasound and specialised stress testing.
- Most symptomatic bridges are managed well with medication, usually beta-blockers or calcium channel blockers. A small, carefully selected group with severe, persistent symptoms may benefit from an operation called surgical unroofing.
- A large 2026 study from Stanford, published in the European Heart Journal, found that for this selected group, surgical unroofing produced lasting relief from angina five years later, with a low complication rate.
You have a muscle bridge. Many people hear that sentence for the first time lying on an angiogram table, nod along, and leave none the wiser. It is rarely explained in much depth, partly because for most people it carries no consequence at all, and partly because the few it does affect need a longer conversation than a results appointment allows.
A myocardial bridge is one of the most common variations in human heart anatomy. The overwhelming majority cause no symptoms, need no treatment, and carry no meaningful risk. For a smaller group of people, though, a bridge can be a real and treatable cause of angina, and telling those two groups apart is what good assessment is for.
This article explains what a muscle bridge actually is, why most are harmless, how the troublesome minority are identified, and what the newest evidence says about treating them.
What a Myocardial Bridge Actually Is
The coronary arteries, the vessels that supply blood to the heart muscle itself, normally run along the outer surface of the heart, sitting on top like rivers on a landscape. A myocardial bridge is a stretch where one of these arteries takes a short detour and tunnels down through the heart muscle before surfacing again on the other side.
The band of muscle lying over the tunnelled segment is the “bridge.” The buried portion of artery is sometimes called the tunnelled segment. It is present from birth, which means a bridge is a variation in how the heart formed rather than something that develops, wears in, or is caused by lifestyle. Around 97% of bridges occur in the same vessel, the left anterior descending artery, which runs down the front of the heart.
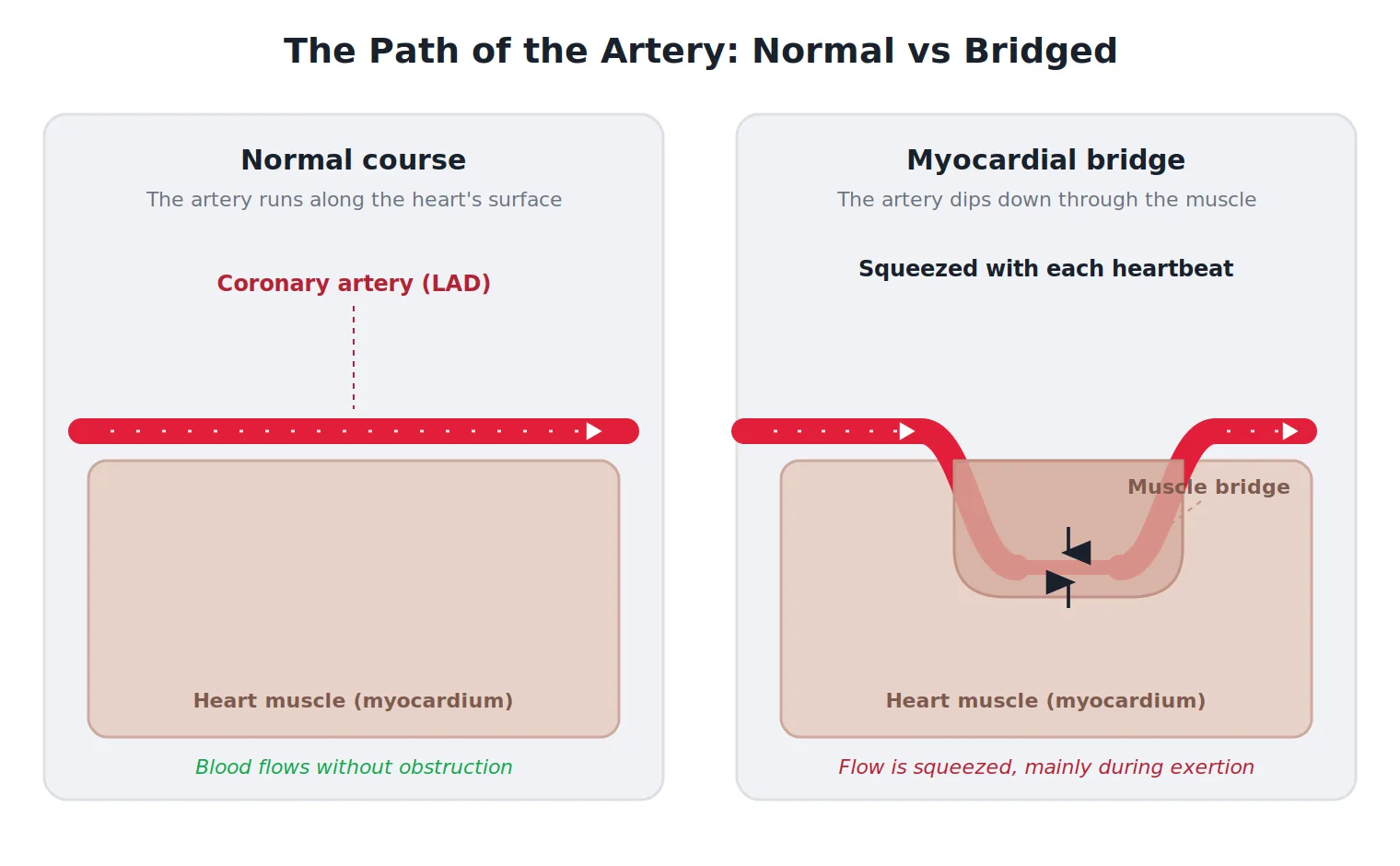
The reason a bridge can matter comes down to mechanics. Every time the heart contracts, the muscle squeezes. If a segment of artery is buried inside that muscle, it gets squeezed too, narrowing with each beat. The heart does most of its own blood supply during the relaxation phase between beats, and in a bridge the squeeze can linger into that phase, slightly delaying the artery from springing back open. When the heart is calm and beating slowly, this rarely matters. When the heart beats fast, during exertion, stress, or strong emotion, the relaxation phase shortens, and the lingering squeeze has proportionally more effect. This is why bridge symptoms, when they occur, are so often tied to exertion.
Why Most Bridges Cause No Trouble
The single most important thing to understand about muscle bridges is how common they are, and how rarely they cause problems. The numbers vary enormously depending on how you look for them, which is itself revealing.
people may have a myocardial bridge when hearts are examined closely, yet only a small fraction ever experience symptoms from it.
Imaging and autopsy studies, summarised in European Heart Journal, 2026
On a standard coronary angiogram, bridges turn up in only a few percent of people. On detailed imaging such as CT coronary angiography, the figure is much higher, and when hearts are examined directly the prevalence rises further still, into the range of one in five to one in four. The gap between these numbers tells the story: most bridges are shallow, short, and functionally silent. They are found incidentally, if they are found at all, and they sit quietly for a lifetime.
So if you have been told you have a bridge and you have no symptoms, the most likely situation by far is that the bridge is an anatomical footnote rather than a problem. It does not need fixing, and in most cases it does not need anything beyond the awareness that it is there.
When a Bridge Does Cause Symptoms
A minority of people with a bridge experience real symptoms, most commonly chest pain or tightness that comes on with exertion or stress and eases with rest. Some describe breathlessness, and some report that episodes last noticeably longer than a typical bout of exertional chest pain. Whether a particular bridge causes symptoms depends on several features working together.
Length of the bridge
A longer tunnelled segment means more of the artery is subject to the muscle’s squeeze with each beat. Longer bridges are more likely to be functionally significant than short ones.
Depth of the tunnel
How deeply the artery is buried in the muscle matters. A deeper bridge is compressed more forcefully, so deeper bridges tend to have a greater effect on blood flow than superficial ones.
Heart rate and exertion
A fast heart rate shortens the relaxation phase when the artery would normally recover. This is why bridge symptoms typically appear during exercise, stress, or strong emotion rather than at rest.
Coronary spasm
Bridges frequently sit alongside a tendency for the artery to spasm, or tighten suddenly. Spasm can drive symptoms independently of the bridge itself and changes how the condition is treated.
Endothelial function
The artery lining near a bridge is often under abnormal mechanical stress, which can impair its ability to relax and widen on demand. This contributes to symptoms in many bridge patients.
Plaque just upstream
The disturbed blood flow around a bridge tends to encourage fatty plaque to build up in the artery just before it, which can add its own contribution to symptoms and risk.
Because several of these factors often coexist, a bridge that looks modest on a picture can still be the source of meaningful symptoms, and a bridge that looks dramatic can sit silent. Anatomy alone does not settle the question. This is the central difficulty in managing bridges, and it shapes everything about how they are investigated.
Why a Bridge Is Harder to Diagnose Than It Sounds
It would be reasonable to assume that if a bridge shows up on an angiogram, the picture tells the whole story. In practice it rarely does. A standard coronary angiogram is good at finding flow-limiting blockages, but it has low sensitivity for bridges and tells you little about how deep a bridge is, how long it is, or whether it is actually responsible for a person’s symptoms.
When symptoms and anatomy do not obviously line up, cardiologists turn to more detailed assessment. Intravascular ultrasound, a tiny ultrasound probe threaded into the artery, is considered the most reliable way to confirm a bridge and measure its length and depth precisely. To judge whether the bridge is functionally significant, meaning it genuinely restricts flow under stress, specialised measurements are taken while the heart is pushed to work harder with a medication called dobutamine. Standard pressure-wire measurements used for ordinary blockages are not adequate for assessing a bridge, which is part of why bridge assessment is a specialised undertaking.
A particular pattern on a stress echocardiogram, a transient buckling in one part of the heart wall with the tip of the heart spared, can also raise suspicion of a bridge non-invasively. The broader point for patients is that confirming a bridge as the cause of symptoms is a deliberate, layered process, not a single glance at an angiogram.
Finding a bridge and proving that the bridge is the cause of someone’s pain are two very different things. Most bridges are bystanders. The clinical skill lies in identifying the minority that are genuinely to blame, and treating that group properly without over-treating everyone else.
Heart Matters editorial
How Symptomatic Bridges Are Treated
Medication comes first
For the large majority of people whose bridge does cause symptoms, medication is the first and usually the only treatment needed. The two mainstays are beta-blockers and calcium channel blockers. Both work in part by slowing the heart and easing the force of each contraction, which lengthens the relaxation phase and reduces the squeezing effect on the bridged artery. They can be very effective, and many people are managed comfortably on them for years.
One counter-intuitive point is worth knowing. Nitrates, which are a standard reliever for ordinary angina, are often used with caution in bridge patients and can sometimes make symptoms worse rather than better. If you have a bridge and find that a nitrate spray does not help your chest pain in the way you would expect, or seems to aggravate it, that is worth mentioning to your cardiologist rather than dismissing.
When an operation is considered
A small, carefully selected group of patients continue to have severe, life-limiting symptoms despite the best medication can offer, and have a bridge that has been confirmed as genuinely significant on detailed testing. For this group, a surgical option exists. The preferred operation is called surgical unroofing, in which the surgeon carefully divides the band of muscle lying over the tunnelled artery, freeing the vessel to return to the surface where it belongs.
Two other approaches, placing a stent inside the bridged segment or performing a coronary artery bypass, have generally proved less satisfactory for bridges specifically, which is why unroofing has become the favoured operation in suitable patients. It is important to stress that this is a treatment for a minority within a minority. The vast majority of people with a bridge will never come close to needing it.
What the Latest Research Shows
Until recently, a fair question about unroofing was whether its benefits lasted. Earlier work had shown good results at six months, but long-term evidence was thin. A 2026 study from Stanford University, published in the European Heart Journal, has now provided the most substantial answer to date.
The researchers followed 218 patients who had undergone surgical unroofing of a significant bridge in the left anterior descending artery after medication had failed to control severe angina. The patients were relatively young, with a median age of 49, and around 60% were women. They were followed for a median of five years.
patients reported a meaningful, lasting improvement in their angina five years after surgical unroofing, with episodes typically falling from weekly to monthly.
Pargaonkar et al., European Heart Journal, 2026
Across every measure of symptoms and quality of life, patients reported significant and durable improvement, and most needed fewer anti-anginal medications afterwards than before. When the surgical patients were compared with a closely matched group who had a similarly significant bridge but were managed with medication alone, the surgical group showed greater relief from physical limitation and from the frequency of their angina. Importantly, the operation proved safe in experienced hands, with a low rate of major cardiac events over the follow-up period and none of those events attributed to the surgery itself.
The practical message for patients is measured rather than dramatic. For the specific, carefully selected group with a confirmed significant bridge and severe symptoms that medication cannot control, surgical unroofing is now backed by solid long-term evidence as a worthwhile option. It is not a treatment for an incidental finding, and it is not a first step. It is a well-supported last resort for those who genuinely need it.
The Bridge Is Rarely the Whole Story
One of the most useful insights from the recent research is that a bridge often travels with company. In the Stanford group, who were of course a highly selected population with difficult symptoms, almost all had a small build-up of fatty plaque in the artery just upstream of the bridge, and the great majority showed evidence of coronary spasm or of an artery lining that did not relax normally on testing.
This matters for two reasons. First, it explains why a few patients still have some symptoms even after a technically successful unroofing: if spasm or lining dysfunction was contributing, removing the muscle band does not address that part of the problem, and ongoing medication may still be needed. Second, the tendency for plaque to form just before a bridge is part of why attention to general cardiovascular risk, including the standard modifiable risk factors, remains relevant even when the headline issue is a structural one present from birth.
Heart Matters Resource
When in Doubt, Get Checked Out
If you have been told you have a muscle bridge and you are experiencing chest pain, breathlessness, or symptoms on exertion, those symptoms are worth a proper conversation with your cardiologist rather than assuming the bridge is harmless.
Conclusion
A myocardial bridge is, for most people, exactly what it usually sounds like when a cardiologist mentions it in passing: a common quirk of heart anatomy that asks nothing of you and changes nothing about your life. If you have one and no symptoms, that is almost certainly where the story ends.
For the minority who do have symptoms, a bridge is a real and treatable condition. The key is careful assessment to confirm whether the bridge is genuinely the cause, sensible use of medication for the many, and a well-evidenced surgical option held in reserve for the few who need it. If you have a bridge and symptoms that affect your daily life, the right next step is not to worry alone but to have it properly evaluated, because the difference between a harmless bystander and a treatable problem is exactly the kind of question modern cardiology is now well equipped to answer.
Related Reading
- Angina: What It Is, What It Feels Like, and How It’s Treated
- Chest Pain: Understanding Symptoms, Causes and Evaluation
- SCAD: Spontaneous Coronary Artery Dissection
- CT Coronary Angiogram (CTCA): What It Is and What It Shows
- Nitrates for Angina: GTN Spray, Patches, and Tablets Explained
- Recovering After a Coronary Angiogram: What to Expect
- Understanding Your Cardiovascular Risk Factors